Want to read offline? [Download PDF]
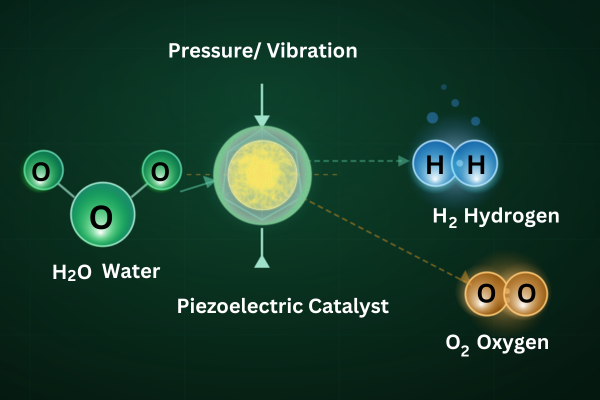
Generating green hydrogen from water splitting is pivotal to the global transition to low-carbon energy systems. Among emerging technologies, piezocatalysis, the conversion of mechanical energy directly into chemical energy via piezoelectric materials, has captured growing attention as a novel, eco-friendly approach to drive water splitting without the need for external electrical input [1] Traditionally, hydrogen production has relied on electrolysis powered by grid electricity or fossil fuels, which limits overall sustainability. Piezocatalytic systems offer a route to harvest ubiquitous mechanical energy sources, including vibrations, fluid motion, and ultrasonic waves, to induce charge separation in materials that can catalyse the water-splitting reaction:
This fundamentally shifts the paradigm by using mechanical deformation of piezoelectric nanomaterials to create internal potentials that facilitate redox reactions at the catalyst surface.[2]
Fundamentals of Piezocatalytic Water Splitting
Piezocatalysis builds on the classical piezoelectric effect: non-centrosymmetric crystalline materials generate separation of positive and negative charges when subjected to mechanical stress. In the context of water splitting, this stress-induced piezopotential drives charge carrier separation and migration, enabling the oxidation of water and reduction of protons at the catalyst interface.[2] Compared with thermal or light-driven methods, piezocatalysis uniquely enables activation using mechanical stimuli alone , potentially harvesting energy from ambient vibrations or engineered mechanical systems. [1]
Key performance factors in piezocatalysis include the piezoelectric coefficient of the material, surface area, defect structures, and charge carrier management. Nanostructured piezocatalysts are particularly effective due to their high surface-to-volume ratio and enhanced contact with water molecules, improving reaction kinetics and hydrogen evolution.
Emerging Material Systems and Performance Benchmarks
Recent research demonstrates that a range of piezoelectric materials, including oxide ceramics, metal-organic frameworks (MOFs), and engineered polymers, can catalyse hydrogen production from water when mechanically activated:
- Ferroelectric BaTiO₃ nanoparticles have delivered hydrogen production rates (>650 μmol g⁻¹ h⁻¹) under ultrasonic vibration, illustrating high piezoelectric conversion efficacy and competitive performance with established catalytic systems.[3]
- BiFeO₃-based composites doped or combined with other semiconductors have shown simultaneous H₂ and H₂O₂ production from water, offering enhanced catalyst redox activity without sacrificial age[4]
- Conjugated microporous polymers with donor–acceptor frameworks demonstrate high H₂ yield rates by harnessing strong internal electric fields and efficient charge separation under mechanical excitation.[5]
- Metal-organic framework nanosheets with engineered piezoresponse exhibit efficient hydrogen evolution by combining high surface area with intrinsic electric fields. [6]
These examples illustrate that carefully designed nanostructured piezocatalysts can rival or complement conventional photocatalytic and electrolysis approaches, with the added benefit of mechanical energy utilisation.
Industry Challenges and Insights
Despite exciting progress, piezocatalytic water splitting is still predominantly at the research and development stage. Practical industry deployment will require:
- High catalyst durability under continuous mechanical cycling
- Scalable synthesis of piezocatalyst nanoparticles
- Reactor designs that effectively couple mechanical input with catalytic surfaces
- Cost-effective manufacturing pathways
Material engineering strategies to address these include defect engineering to increase polarization intensity and charge carrier lifetimes, heterostructure formation to facilitate electron–hole separation, and size control to enhance active surface sites. Integration with complementary catalytic techniques such as piezo-photocatalysis further amplifies performance by combining mechanical and solar energy harvesting.
Why Choose Nikalyte Nanoparticle Source for Piezocatalyst Nanoparticles
At the core of effective piezocatalyst design is the synthesis of high-quality nanoparticles with precise control over size, composition, and surface properties. Nikalyte’s NL-UHV Nanoparticle Deposition Source provides industry-leading capabilities for producing pure, non-agglomerated nanoparticles in an ultra-high vacuum environment with inline mass-filtered size control. These sources enable deposition of nanoparticles ranging from 1–20 nm with tunable structure and composition, crucial for optimising piezocatalytic performance in water splitting systems. The ability to integrate with existing PVD vacuum systems or be customised for bespoke deposition setups makes them a versatile tool for advanced catalyst engineering.
This precision translates directly to improved catalytic performance: narrower particle size distributions increase active surface area and reactivity, while controlled alloying and composition improve piezopotential generation and charge transfer properties critical to high-efficiency hydrogen evolution.
Conclusion
Piezocatalytic water splitting represents a transformative approach to green hydrogen production, turning ambient or engineered mechanical energy into chemical fuel with minimal carbon footprint. Supported by advances in nanostructured materials and deposition technologies, the field is moving toward real-world implementation. Strategic material engineering and scalable manufacturing will be central to bridging the gap between promising lab results and industrial solutions.
Contact us to explore how our nanoparticle deposition sources can accelerate your catalyst development, empower scalable manufacturing, for clean hydrogen production.
References
- Xu, X., Wang, Y., Cheng, W., Zhai, H., Xiao, L., Qin, L., & Chen, D. (2024). Recent advances in piezocatalytic hydrogen production and prospects. Surfaces and Interfaces, 54, 105245. https://doi.org/10.1016/j.surfin.2024.105245
- Li, S., Zhang, X., Yang, F., Zhang, J., Shi, W., & Rosei, F. (2024). Mechanically driven water splitting over piezoelectric nanomaterials. Chem Catalysis, 4(2), 100901. https://doi.org/10.1016/j.checat.2024.100901
- Su, R., Hsain, H. A., Wu, M., Zhang, D., Hu, X., Wang, Z., Wang, X., Li, F. T., Chen, X., Zhu, L., Yang, Y., Yang, Y., Lou, X., & Pennycook, S. J. (2019). Nano-ferroelectric for high efficiency overall water splitting under ultrasonic vibration. Angewandte Chemie International Edition, 58(42), 15076–15081. https://doi.org/10.1002/anie.201907695
- Zeng, H., Liu, C., Lan, B., Tan, M., Yu, C., Su, Y., Qiao, L., & Bai, Y. (2024). Bifunctional Bi₀.₉₈Sm₀.₀₂FeO₃/g-C₃N₄ piezocatalyst for simultaneous H₂ and H₂O₂ production. ACS Applied Materials & Interfaces, 16(51), 70566–70574. https://doi.org/10.1021/acsami.4c15127
- Chen, J.-P., Xie, L.-F., Huang, W.-H., Niu, L., Ni, Q.-L., Huang, T.-H., Gui, L.-C., & Wang, X.-J. (2024). Piezocatalytic performances of conjugated microporous polymers with donor–acceptor structures for overall water splitting. ACS Catalysis, 14(10), 7853–7866. https://doi.org/10.1021/acscatal.4c00918
- Zhao, S., Liu, M., Zhang, Y., Zhao, Z., Zhang, Q., Mu, Z., Long, Y., Jiang, Y., Liu, Y., Zhang, J., Li, S., Zhang, X., & Zhang, Z. (2022). Harvesting mechanical energy for hydrogen generation by piezoelectric metal–organic frameworks. Materials Horizons, 9(7), 1978–1983. https://doi.org/10.1039/d1mh01973b
- Nikalyte Ltd. (2025). Nanoparticle deposition source NL-UHV for controlled nanoparticle synthesis. https://www.nikalyte.com/nanoparticle-deposition-source-nl-uhv/